welcome to our website!
research interest
Using cellular resolution recording techniques, we address fundamental questions concerned with the formation, initiation, and progression of epileptic seizures in the intact brain. Epileptic seizures likely arise through the aberrant interaction of local neural populations whose underlying circuitry is not well understood. Despite >100 years of research, even the most basic aspects of epilepsy have escaped our understanding, and up to 30% of patients remain unresponsive to pharmacotherapy. No clear answer exists as to how an epileptic network forms over time, how individual seizures start or terminate, why seizures remain focal in one case, or spread in another. One reason for this persistent lack of understanding has been the technical difficulty to study densely packed epileptic networks at sufficient temporal and spatial resolution. During his post-doc at Columbia University, Dr. Wenzel studied acute focal seizure formation and spread at cellular resolution within and across cortical layers in vivo. His newly formed group at Bonn University combines cutting edge cellular scale chronic in vivo calcium imaging with electrophysiology, and behavioral assessment in various models of chronic epilepsy in mice. The core goal of the group is the identification of basic local circuit elements of chronically seizure producing networks that in the best case are shared across different types of epilepsy.
Experimental techniques

Two-photon in vivo imaging of seizure microprogression
Local cortical network recruitment patterns during seizure microprogression
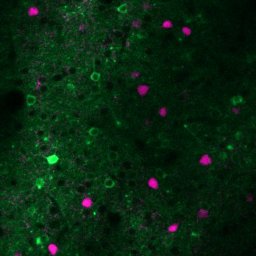
Cellular scale in vivo imaging of neural subtypes
green: Pyramidal cells (PYR) magenta: PV interneurons
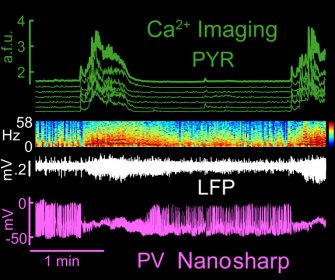
Multimodal recordings across anatomical scales
in vivo
Two-photon Ca2+ imaging +
Local field potential recordings + Intracellular nanosharp recordings
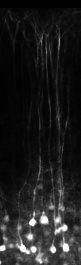

In vivo imaging across cortical layers and in hippocampus
Glass microprism-, or micro-cylinder-assisted multilayer or hippocampal imaging
LAB MEMBERS
Michael Wenzel, M.D. Ph.D., Principal Investigator
2024 – Professorship, Epileptic Micronetworks & Network Neurophysiology, Bonn
2022 – Board certification Clinical Neurology / Habilitation Exp. Neurosciences
2020 – Hertie Excellence Network in Clinical Neuroscience
2018 – Dept. of Epileptology, University Medical Center (UKB) Bonn
2014 – 2018 Post-doc with Rafael Yuste, Columbia University, New York
2011 – 2013 Dept. of Neurology, LMU, Clinical training in Neurology
Mayan Baues, M.S., Ph.D. Student
2020 M.S. in Neuroscience, University of Cologne, Germany
2018 B.S. in Biology, University of Bonn, Germany
Bence Mitlasóczki, M.S., Ph.D. Student
2020 M.S. in Physics, University of Bonn, Germany
2017 B.S. in Physics, University of Szeged, Hungary
Nele Neumann, Technician
2024 B.S. in Pharm. Chemistry, University of Technology, Arts & Science, Cologne
Ahmed Elgokha, Ph.D., Senior Research Associate
2001 B.S. in Chemistry, Menoufia University, Egypt
2005 M.S. in Organic Chemistry, Menoufia University, Egypt
2011 Ph.D. in Organic Chemistry, University of Ulm, Germany
Georgia Schweigert-Bastos, Post-doc
2019 B.S. in Biotechnology, Pennsylvania State University, USA
2021 M.S. in Neuroscience, Georgia State University, USA
2024 Ph.D. in Neuroscience, Georgia State University, USA
Email: Georgia.Schweigert-Bastos@ukbonn.de
Midia Kamali, Ph.D., Research Associate
2011 Doctorate in Veterinary Medicine (DVM), Urmia Azad University, Iran
2018 Ph.D. in Physiology, University of Tehran, Iran
Mai My Hong Nguyen, M.S., Research Associate
2015/2016 Undergraduate Studies, Ho Chi Minh City University, Vietnam
2021 Pharm. D., University of Debrecen, Hungary
2024 M.S. in Neuroscience, University of Bonn, Germany
Merlin Felix Schwering-Sohnrey, B.S., Medical Student
Since 2020 Medical School, University of Bonn, Germany
2020 B.S. in Chemistry, University of Bonn, Germany









Collaborators
Dr. Heinz Beck (In vivo two-photon imaging, experimental epileptology)
Institute for Experimental Epileptology and Cognition Research, Bonn University, Germany
Dr. Dr. Florian Mormann (Single unit recordings in humans)
Department of Epileptology, Bonn University, Germany
Dr. Laura Ewell (Hippocampal memory coding, experimental epileptology)
Institute for Experimental Epileptology and Cognition Research, UC Irvine, USA
Dr. Tony Kelly (Hippocampal dendritic integration, experimental epileptology)
Institute for Experimental Epileptology and Cognition Research, Bonn University, Germany
Dr. Darcy Peterka (In vivo two-photon imaging)
Zuckerman Mind Brain Behavior Institute, Columbia University, New York, USA
Dr. Krishna Jayant (Nanoelectronics, biomedical engineering)
Weldon School of Biomedical Engineering, Purdue University, Indiana, USA
Dr. Yuki Bando (Molecular Biology, Voltage Imaging)
Hamamatsu University School of Medicine, Hamamatsu, Japan
publications
Masala M, Mittag M, Giovannetti EA, O’Neil DA, Distler F, Rupprecht P, Helmchen F, Yuste R, Fuhrmann M, Beck H, Wenzel M*, Tony Kelly*. Aberrant hippocampal Ca2+ micro-waves following synapsin-dependent adenoviral expression of Ca2+ indicators. eLife (2024) https://doi.org/10.7554/eLife.93804.2.sa3
Wenzel M, Racz A, Surges R. Epilepsietypische Hirnaktivität (2023), Chapter 7 in Klinische Elektroenzephalographie (eds Zschocke S, Hansen HC), Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-662-63267-3_7
Wenzel M*, Huberfeld G*, Grayden DB, de Curtis M, Trevelyan AJ. A debate on the neuronal origin of focal seizures. Epilepsia (2023) doi:10.1111/epi.17650
Wenzel M, Bender R, Surges R. From prolonged febrile seizures to epilepsy: potential contributions of HCN channels, Chapter 14 in Febrile Seizures (Baram, Shinnar and Stafstrom) (2023) doi.org/10.1016/B978-0-323-89932-1.00020-2
Bando Y, Wenzel M, Yuste R. Simultaneous two-photon imaging of action potentials and subthreshold inputs in vivo. Nature Communications (2021) 12, 7229
Wenzel M, Leunig A, Han S, Peterka DS, Yuste R. Prolonged anesthesia changes brain synaptic architecture. PNAS (2021) 188 (7) e2023676118
Wenzel M and Hamm JP. Identification and quantification of neuronal ensembles in optical imaging experiments. Journal of Neuroscience Methods (2020) doi.org/10.1016/j.jneumeth.2020.109046
Wenzel M, Hamm JP, Peterka DS, Yuste R. Acute focal seizures start as local synchronizations of neuronal ensembles. Journal of Neuroscience (2019) 39 (43) 8562-8575
Wenzel M*, Han S*, Smith EH*, Hoel E, Greger B, House PA, Yuste R. Reduced repertoire of cortical microstates and neuronal ensembles in medically induced loss of consciousness. Cell Systems (2019) 8, 467–474; *equal contribution
Jayant K*, Wenzel M*, Hamm JP, Bando Y, Sahin O, Shepard KL, Yuste R. Flexible nanopipettes for minimally invasive intracellular electrophysiology in vivo. Cell Reports (2019) 26, 266–278; *equal contribution
Liou JY*, Ma H*, Wenzel M, Zhao M, Baird-Daniel E, Smith EH, Daniel AGS, Emerson R, Yuste R, Schwartz TH, Schevon CA. Role of inhibitory control in modulating focal seizure spread. Brain (2018) 141, Issue 7, 2083–2097; *equal contr.
Wenzel M, Hamm JP, Peterka DS, Yuste R. Reliable and elastic propagation of cortical seizures. Cell Reports (2017) 19:2681–2693; Cover Story
Baird-Daniel E*, Daniel AGS*, Wenzel M, Li D, Liou JY, Laffont P, Zhao M, Yuste R, Ma H, Schwartz TH. Glial waves are triggered by seizure activity and not essential for initiating ictal onset or neurovascular coupling. Cerebral Cortex (2017) 27:3318-3330; *equal contribution
Ricci S, Grandgirard D, Wenzel M, Braccini T, Salvatore P, Oggioni MR, Leib SL, Koedel U. Inhibition of matrix metalloproteinases attenuates brain damage in experimental meningococcal meningitis. BMC Infect Dis. (2014) 14:726
Wenzel M, Jakob L, Wieser A, Schauber J, Dimitriadis K, Schubert S, Pfister HW. Corticosteroid-Induced Meningococcal Meningitis in a Patient With Chronic Meningococcemia. JAMA Dermatology (2014)150(7):752-755
Heinrich D, Wenzel M, Dimitriadis K, Mühlstädt M. An international medical curriculum – first steps of implementation. Medical Education (2013) 47(5):516
Höhne C, Wenzel M, Angele B, Hammerschmidt S, Häcker H, Klein M, Bierhaus A, Sperandio M, Pfister HW, Koedel U. HMGB1 prolongs inflammation and worsens disease in experimental pneumococcal meningitis. Brain (2013) 136:1746-59
Wenzel M, Wunderlich M, Besch R, Poeck H, Willms S, Schwantes A, Kremer M, Sutter G, Endres S, Schmidt A, Rothenfusser S. Cytosolic DNA triggers mitochondrial apoptosis via DNA damage signaling proteins independently of AIM2 and RNA polymerase III. J Immunology (2012) 188(1):394-403
Schmidt A, Schwerd T, Hamm W, Hellmuth JC, Cui S, Wenzel M, Hoffmann FS, Michallet MC, Besch R, Hopfner KP, Endres S, Rothenfusser S. 5′-triphosphate RNA requires base-paired structures to activate antiviral signaling via RIG-I. PNAS (2009) 106(29):12067-72
Funding







Contact
PD Dr. Michael Wenzel
University Hospital Bonn
Venusberg-Campus 1
Building 83
D-53127 Bonn
Germany

